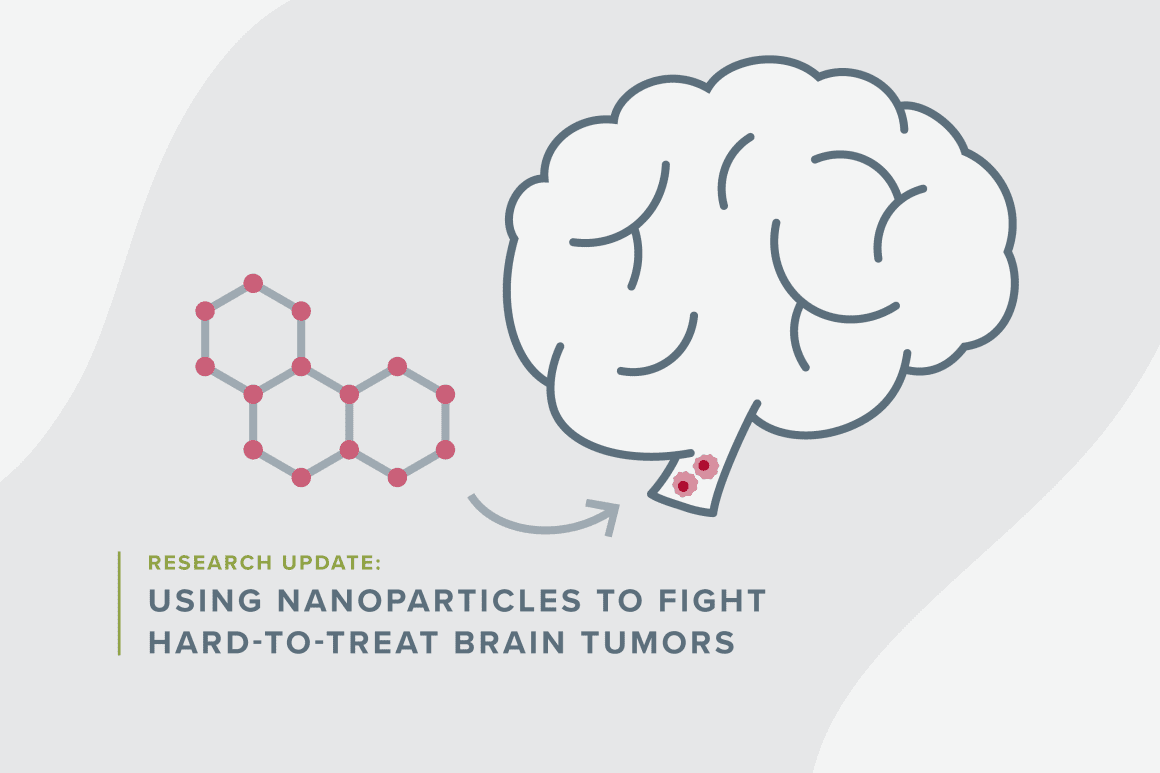
Diffuse intrinsic pontine glioma (DIPG), a rare and currently incurable brain cancer, is so difficult to remove that conventional brain surgery isn’t an option. The tumor cells lodge in the brainstem, weaving around critical nerve cells and other necessary parts of the brain.
But pediatric neurosurgeon Peter Chiarelli, MD, PhD, who leads the Nanoscaled Neuro-technology Research Group at Children’s Hospital Los Angeles, is determined to find other innovative ways to defeat the disease. With support from an Accelerating Impact for Hard-to-Treat Cancer Award from Children’s Cancer Research Fund (CCRF), Chiarelli is using nanoparticles to develop a smarter radiation treatment for DIPG — essentially using microscopic “magnifying lenses” to remove what a surgeon’s scalpel can’t. If effective, the approach could be life-changing and might eventually lead to a cure for the approximately 300 children in the United States who are diagnosed with DIPG each year.
Chiarelli estimates that 200-plus clinical trials have tried to improve outcomes for DIPG. “Survival is still approximately 11 months. And it was 11 months in the 1970s,” he said.
A smarter radiation approach
Radiation is the current standard of care for DIPG, and it can shrink the tumor slightly and help relieve kids’ symptoms for 2-4 months before the tumor regrows. But Chiarelli hopes to boost the effectiveness of radiation by harnessing light-matter interactions to make radiation cell-specific. That would allow doctors to increase the potency of radiation while minimizing harm to healthy brain cells, potentially improving overall survival and reducing side effects.
Nanoparticles are suited well for that purpose. “There are known ways to send nanoparticles, which are small clusters of atoms that have been coded to make them safe and biocompatible, to go where you want them to go,” he noted. His team is using nanoparticles made of iron oxide, a material that has already been approved by the U.S. Food and Drug Administration for other medical uses.
Chiarelli’s team is leveraging a light-matter interaction called the Auger effect, which causes atoms to eject electrons when exposed to light like an X-ray beam. Researchers first inject nanoparticles engineered to target DIPG tumor cells in the brainstem. Then they treat the area with radiation. Instead of the cells simply absorbing the radiation, as normally happens, the atoms in the nanoparticles eject electrons when hit by the light. The confined action within the tumor cell acts like a smart bomb, killing the tumor cell while preserving nearby healthy cells, Chiarelli explained.
Improving survival
Chiarelli recently published research showing the success of a similar treatment in adult glioblastoma. That study showed that combining radiation with iron oxide nanoparticles significantly reduced tumor growth and doubled the survival time when tested in animal models. Now, he hopes that the approach is just as promising for hard-to-treat pediatric brainstem tumors — the goal is to increase periods of remission and improve quality of life for patients with DIPG, with a longer-term goal of developing a refined therapy strategy that leads to a cure. If the pre-clinical testing goes well, the treatment could be ready for a pediatric clinical trial in 4-5 years.
Chiarelli believes the “smart radiation” approach could be used for many other cancers. But fulfilling that vision depends on critical early-stage research, which would not be possible without philanthropic support.
“CCRF’s assistance and their belief in us has been a major stepping stone,” he said. “We really want to make a dent in one of the hardest brain tumors there is to treat. That, overall, is a fairly challenging situation, but I think we’re set up for success.”
Your donation supports researchers like Dr. Chiarelli.
Your support propels bold ideas forward and empowers researchers to discover treatments that are better and safer for kids, and ensure every child can have a long, healthy life after cancer.